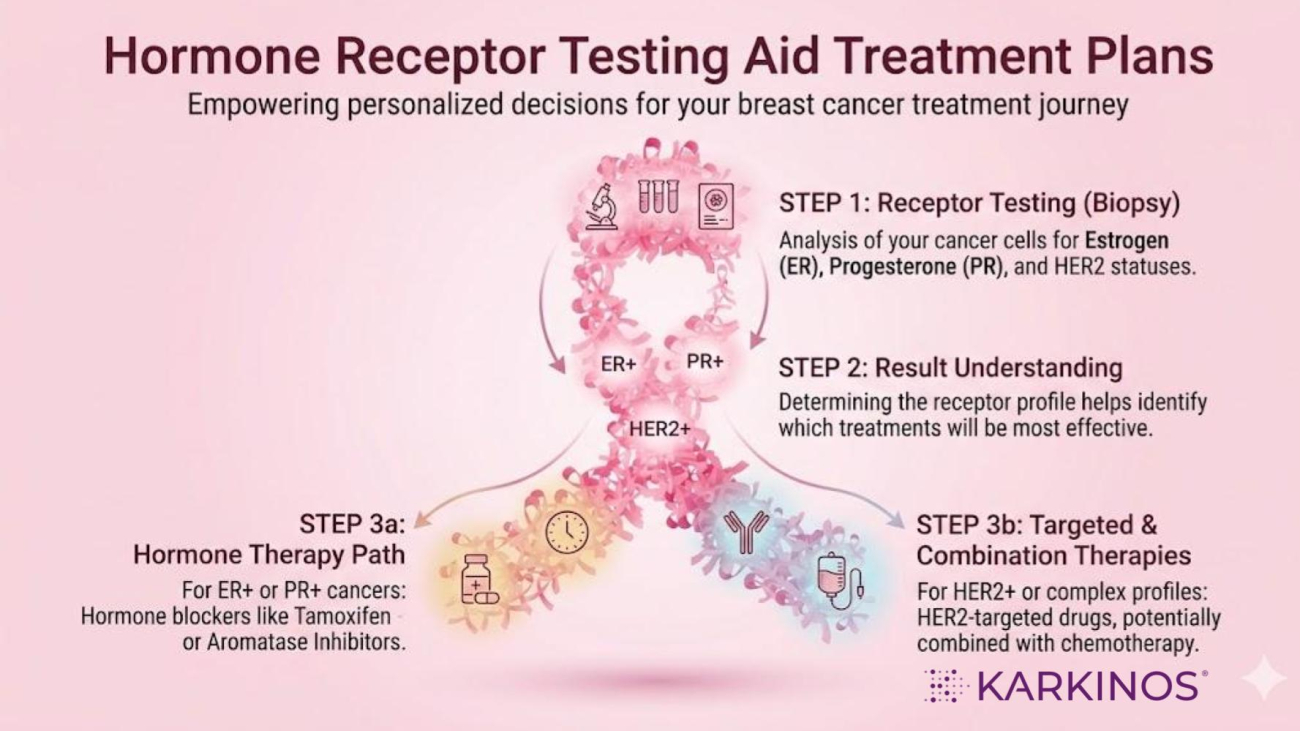
Hormone receptor testing determines whether breast cancer cells express Estrogen Receptor and Progesterone Receptor, helping identify tumours that depend on hormonal signalling for growth. This information is critical because it predicts response to endocrine therapy and plays a central role in guiding treatment decisions and prognosis.
In our previous blog on oestrogen and progesterone, we understood that luminal epithelial cells lining the breast ducts and lobules are naturally equipped with oestrogen receptors (ER) and progesterone receptors (PR), which act as molecular switches that govern cell growth across the menstrual cycle, pregnancy, and menopause.
We now know that oestrogen drives cell proliferation, progesterone refines and stabilises that growth, and that most breast cancers arise from hormone-responsive cells that retain the same receptor machinery found in normal tissue.
We also introduced the molecular subtypes and mapped receptor status to treatment strategy in broad terms. What we have not yet explored is the question that follows every breast cancer diagnosis: how do doctors actually measure receptor activity, what do the numbers on a pathology report mean, and how precisely does receptor status shape treatment decisions? This article answers those questions directly.
Why the Receptor Exists and What Goes Wrong
The oestrogen receptor is not a disease marker. It is a normal, essential component of breast cell biology. Every cell in the body carries a full set of DNA, but which genes get switched on depends on chemical signals arriving from outside the cell. Oestrogen is one of those signals, and the oestrogen receptor is the protein that reads it.
The receptor is encoded by a gene called ESR1, located on chromosome 6. It functions as a ligand-dependent transcription factor, a protein that, once oestrogen binds to it, travels into the cell nucleus and activates specific genes that control growth, division, and differentiation.
One end of the protein attaches to oestrogen, the middle grips DNA, and the other end switches the target gene on. This machinery is entirely appropriate in normal breast tissue, where oestrogen signalling drives ductal development during puberty, cyclical remodelling during menstruation, and dramatic tissue expansion during pregnancy.
In ER-positive breast cancer, this system is not broken; it is hijacked. The cancer cell retains its receptors and continues using oestrogen as a growth signal, but without the normal checks that limit how much it divides, leading to uncontrolled growth. This is actually good news for treatment: a cancer that relies on a specific hormone has shown a weakness that can be targeted.
|
The progesterone receptor (PR) works in close coordination with ER. Importantly, the gene that codes for PR is itself switched on by oestrogen signalling. When ER is active, it induces PR expression. This is why more than 80% of ER-positive breast cancers are also PR-positive.
A tumour that is ER-positive but PR-negative is therefore significant: it suggests that the oestrogen receptor is present, but its downstream signalling is partially disrupted. As we will see, this distinction directly influences how a cancer is classified and treated.
|
How Pathologists Measure Receptor Status: The IHC Test
The test used to detect ER and PR in tumour tissue is called immunohistochemistry, or IHC. A chemical antibody is applied to a thin slice of tumour tissue from a biopsy. This antibody is designed to bind specifically to ER or PR protein. Wherever the protein is present in cell nuclei, a brown stain appears. The pathologist then examines the slide under a microscope and quantifies what they see.
IHC is the global standard for receptor testing because it can be performed on standard paraffin-embedded tumour blocks, is cost-effective, and is widely available across Indian hospitals and pathology laboratories.
The key question the test answers is not simply whether receptors are present, but in how many cells and at what intensity, because both matter for prognosis and treatment response.
|
Reading the Score: Allred and H-Score Explained
Simply saying “positive” or “negative” doesn’t tell the whole story. Instead of an on-off switch, think of hormone receptors like a dimmer switch. Some cancers are only slightly lit up by hormones, while others shine bright.
To measure this, pathologists use two main scoring systems. These numbers help the doctor decide exactly how much hormone therapy is needed.
1. The Allred Score (Total: 0 to 8)
The Allred score is like a two-part grade. It looks at how many cells are affected and how strong the hormone signal is.
- Step A (The Percentage): The pathologist looks at 100 cells. If 0 cells have receptors, the score is 0. If nearly all 100 cells have them, the score is 5.
- Step B (The Intensity): How “dark” or strong is the stain? 1 is weak, 2 is intermediate, and 3 is strong.
The Result: Doctors add these two numbers together.
- 0 to 2: Considered Negative.
- 3 to 8: Considered Positive.
A score of 8/8 is the highest possible. It means almost every cancer cell is listening to hormones, and the signal is very loud. These cancers usually respond very well to hormone-blocking pills.
2. The H-Score (Total: 0 to 300)
The H-Score is more detailed. It assigns a specific weight to the cells based on the strength of receptor presence.
- Low H-Score (under 100): The cancer is low positive. It uses some hormones, but it might be growing for other reasons, too.
- High H-Score (200 to 300): The cancer is high positive. It is very dependent on hormones.
Why the Exact Number Matters
If the report just said positive, it would not indicate whether the score was an Allred 3 or an Allred 8.
- If the score is very high: Hormone therapy (like Tamoxifen) will likely be the most powerful tool.
- If the score is low but still positive: The cancer is only partially driven by hormones. In these cases, the oncologist might suggest combining hormone therapy with other treatments, like chemotherapy, to ensure all the cancer cells are targeted.
From Receptor Status to Molecular Subtype
ER and PR status are not interpreted in isolation. They are part of a four-marker panel — ER, PR, HER2, and Ki67 — that together place a breast cancer into a molecular subtype. This classification is critical because the subtypes differ substantially in behaviour, prognosis, and the treatment they require.
Luminal A is the most common and most favourable subtype: ER-positive, PR high, HER2-negative, and Ki67 low (below 14%). These tumours grow slowly, resemble normal breast cells histologically, and typically respond well to endocrine (hormone) therapy alone. Chemotherapy is often not required.
Luminal B (HER2-negative) is ER-positive but PR low or absent, HER2-negative, and Ki67 high (above 20%). These tumours grow faster, have a worse prognosis than Luminal A, and usually require endocrine therapy combined with chemotherapy.
Luminal B (HER2-positive) is ER-positive, HER2 overexpressed, with any PR and any Ki67. These cancers require endocrine therapy, anti-HER2 therapy such as trastuzumab, and usually chemotherapy.
The practical implication for patients: a PR-negative result in an otherwise ER-positive tumour is not a minor detail. It typically shifts the classification from Luminal A to Luminal B and changes the treatment plan.
Recent research indicates that using a combined endocrine receptor score, which averages the expression of estrogen receptors (ER) and progesterone receptors (PR), is a more effective predictor of disease-free survival than evaluating each marker separately. This reinforces the importance of not treating PR as an afterthought in the report.
The Hormonal Risk Factors: Why Biology Matters Before Diagnosis
ER-positive breast cancer is not only treated through the oestrogen-receptor axis, but it is also caused, in large part, through it. As we explored in the previous article, oestrogen drives cell proliferation in breast tissue. The longer and more often breast cells are stimulated by oestrogen over a lifetime, the greater the cumulative opportunity for DNA replication errors. The risk factors for ER-positive, luminal breast cancer specifically track this biology closely.
Reproductive milestones that increase lifetime oestrogen exposure increase risk: menarche before age 12, first pregnancy after age 30, never having been pregnant (nulliparity), and menopause after age 55. The relationship between these factors and breast cancer subtype is not uniform; the link is substantially stronger for hormone receptor-positive, luminal subtypes than for triple-negative breast cancer, which is a biologically different disease with different drivers.
The Indian context adds important nuances that are directly relevant to readers in this country. Epidemiological data show that the age-standardised incidence rate of breast cancer in India is rising, driven by shifts including later age at first birth, lower parity, shorter breastfeeding duration, rising rates of overweight and obesity, and changing dietary and physical activity patterns.
These changes — largely markers of urbanisation — are pushing the hormonal risk profile of Indian women closer to patterns seen in higher-incidence Western populations. Studies in Indian women confirm that both early menarche and late menopause increase breast cancer risk by extending the number of ovulatory cycles and total oestrogen exposure, and that parity and breastfeeding are specifically associated with reduced risk of the luminal subtype.
One epidemiological feature distinguishes India from Western populations and is clinically important: while ER/PR-positive cancers remain the most common subtype in India at approximately 50–60% of cases, the proportion of triple-negative breast cancer is substantially higher in Indian patients — approximately 25–30% — compared to Western figures of around 10–15%. This means that both luminal and triple-negative disease require active clinical and public health attention in India, and that Western risk models and treatment guidelines should be applied with awareness of this difference.
What ER/PR Status Means for Treatment
Once receptor status is established, the treatment rationale is straightforward: if this cancer requires oestrogen to grow, interrupt the oestrogen signal.
Selective oestrogen receptor modulators (SERMs), principally tamoxifen, act like a key that jams the lock — occupying the receptor so that real oestrogen cannot bind. Tamoxifen is effective in both premenopausal and postmenopausal women, significantly reduces recurrence risk over 5 years of treatment, and further reduces risk when extended to 10 years. It is widely available and affordable across India, making it the backbone of adjuvant endocrine therapy for many patients.
Aromatase inhibitors (AIs) — anastrozole, letrozole, and exemestane — work upstream of the receptor by blocking the enzyme aromatase, which converts androgens into oestrogen in peripheral tissues such as fat and muscle. After menopause, peripheral conversion is the main source of oestrogen in the body, so AIs are particularly effective in postmenopausal women. They have demonstrated superior efficacy compared to tamoxifen in this group. For premenopausal women at intermediate or high recurrence risk, the combination of ovarian suppression and an AI is increasingly the recommended standard.
Selective oestrogen receptor degraders (SERDs), such as fulvestrant, go further. They bind to the receptor and mark it for complete destruction, rather than merely blocking it. These are used mainly in the metastatic setting, particularly when resistance to earlier endocrine therapies has developed.
An important clinical caveat: a subset of ER-positive cancers acquire resistance to endocrine therapy over time, particularly after prolonged aromatase inhibitor use. The most common mechanism involves mutations in the ligand-binding domain of the ESR1 gene — at positions Y537 and D538 — that allow the receptor to remain constitutively active even in the absence of oestrogen. Liquid biopsy techniques can now detect these mutations in circulating tumour DNA, informing decisions about escalating or changing therapy. This is an active area of clinical development, both globally and in India.
References
Mohammed H et al. The combined endocrine receptor in breast cancer. British Journal of Cancer, 2016. https://www.nature.com/articles/bjc2016206
Öztürk Ç et al. Reporting Hormone Receptor Expression in Breast Carcinomas. Applied Immunohistochemistry & Molecular Morphology, 2024. https://journals.sagepub.com/doi/10.1177/10668969241265068
Madabhushi A et al. Allred Scoring of ER-IHC Stained Whole-Slide Images. Diagnostics (MDPI), 2022. https://www.mdpi.com/2075-4418/12/12/3093
Li JJX, Tse GM. Hormone receptors. PathologyOutlines.com. https://www.pathologyoutlines.com/topic/breastmalignanthormonereceptors.html
Jeselsohn R et al. ESR1 activating mutations: From structure to clinical application. Biochimica et Biophysica Acta, 2022. https://www.sciencedirect.com/science/article/pii/S0304419X2200155X
Merenbakh-Lamin K et al. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. PMC, 2013. https://pmc.ncbi.nlm.nih.gov/articles/PMC3903423/
Breast Cancer Research Foundation. Molecular Subtypes of Breast Cancer. https://www.bcrf.org/about-breast-cancer/molecular-subtypes-breast-cancer/
Cheang MCU et al. Ki67 index, HER2 status, and prognosis of luminal B breast cancer. JNCI, 2009. https://pubmed.ncbi.nlm.nih.gov/19436038/
Thangaraju P, Venkatesan S. Menopause Hormone Therapy and Breast Cancer in India. PMC, 2021. https://pmc.ncbi.nlm.nih.gov/articles/PMC7978056/
Das U, Soren S, Kar N. Menstrual and reproductive factors in Indian women with breast cancer. Archives of Public Health, 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11041000/
Early menarche, late menopause and breast cancer subtypes: a scoping review. PMC, 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11460410/
Bose S et al. Genomic hallmarks of endocrine therapy resistance in ER/PR+ breast tumours. Communications Biology, 2025. https://www.nature.com/articles/s42003-025-07606-x
Napoli M et al. Current medical treatment of ER-positive breast cancer. PMC, 2015. https://pmc.ncbi.nlm.nih.gov/articles/PMC4549764/
Espinosa E et al. An emerging generation of endocrine therapies in breast cancer. npj Breast Cancer, 2023. https://www.nature.com/articles/s41523-023-00523-4
NCI. Hormone Therapy for Breast Cancer. https://www.cancer.gov/types/breast/breast-hormone-therapy-fact-sheet
Medical disclaimer: This article is for general educational purposes only and does not constitute medical advice. Guidelines vary by centre, patient profile, and resource availability. Always consult a qualified oncologist or breast cancer specialist for individual guidance.
Karkinos Healthcare